De-escalation strategies of dual antiplatelet therapy in patients undergoing percutaneous coronary intervention for acute coronary syndrome
Article information
Abstract
Antiplatelet therapy is important for reducing systemic and local thrombotic events in patients undergoing percutaneous coronary intervention (PCI) for acute coronary syndrome (ACS). Antiplatelet treatment regimens, along with dual antiplatelet therapy consisting of aspirin and a P2Y12 inhibitor for patients receiving PCI, have frequently changed over the years. With improvements in the understanding of the prognostic relevance of bleeding events in patients with PCI, as well as the safety and efficacy of drug-eluting stents, several randomized controlled trials (RCTs) have been conducted on antiplatelet treatment strategies associated with a more favorable balance between ischemic and bleeding risks. Several key RCTs for appropriate antiplatelet therapy in patients receiving PCI for ACS have been reported, and practical guidelines have been updated. This manuscript presents the results of major RCTs on de-escalation strategies of dual antiplatelet treatment in patients receiving PCI for ACS.
INTRODUCTION
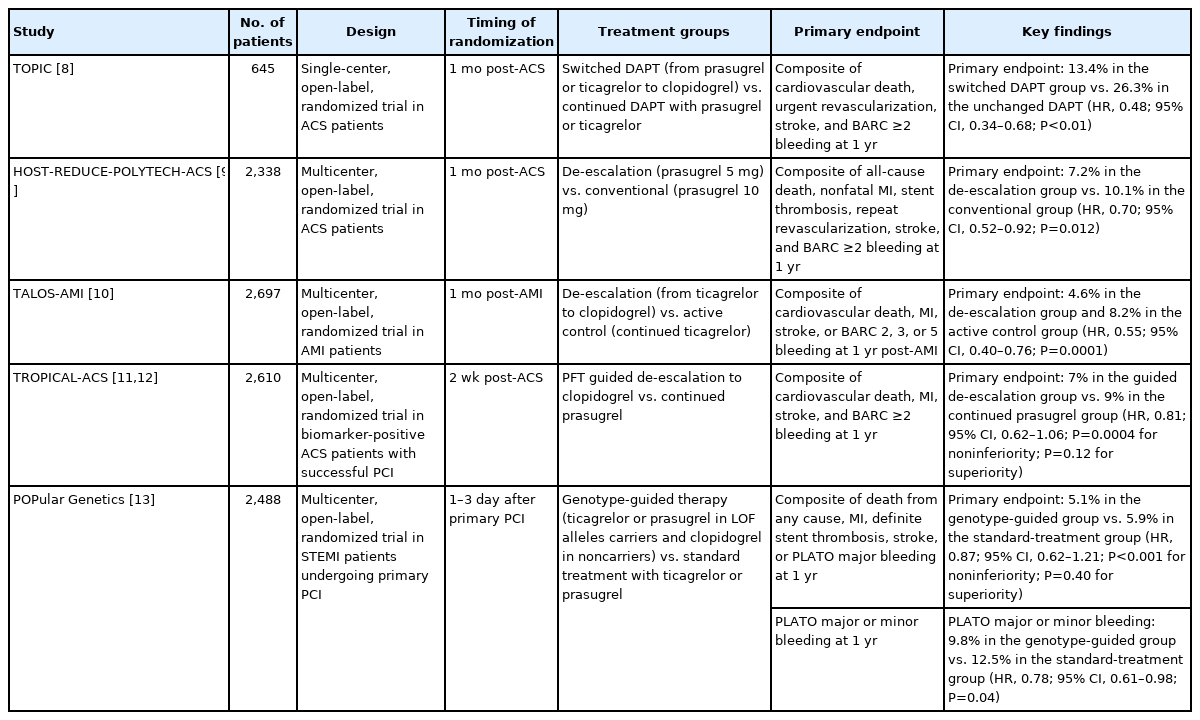
Antithrombotic therapy is fundamental for preventing thrombotic events in patients with acute coronary syndrome (ACS), particularly those undergoing percutaneous coronary intervention (PCI) [1]. Over the past few decades, advances have been made in antithrombotic therapies developed to alleviate the risk of systemic and local thrombotic events. The current standard antiplatelet therapy after PCI with drug-eluting stents (DESs) is dual antiplatelet therapy (DAPT) with aspirin and a P2Y12 inhibitor [1,2]. However, prolonged and potent DAPT increases the risk of bleeding, which offsets the benefit from reducing recurrent ischemic events [3–5]. Newer-generation DESs require less aggressive antithrombotic drugs to prevent ischemic events. Furthermore, there is increasing recognition of the prognostic relevance of bleeding events in patients undergoing PCI [6,7]. For these reasons, several key randomized controlled trials (RCTs) have been recently conducted to identify antiplatelet therapies associated with a more favorable balance between ischemic and bleeding risks (Table 1) [8–13]. This review presents newer evidence regarding de-escalation strategies of DAPT for ACS patients receiving PCI, focusing on recent clinical trials and their implications.
DE-ESCALATION STRATEGIES
Potent P2Y12 inhibitors, such as prasugrel and ticagrelor, show stronger and more consistent platelet inhibition and have been shown to reduce major adverse cardiovascular events when compared to clopidogrel [4,5]. Therefore, current guidelines support the preferred use of prasugrel and ticagrelor over clopidogrel due to their significant ischemic benefit in patients with ACS [1,14]. However, these potent P2Y12 inhibitors carry a higher risk of bleeding [15]. Considering that the risk of ischemic events after PCI for ACS is more prominent in the early phase, de-escalation after short-term DAPT with a potent P2Y12 inhibitor could achieve an optimal balance between ischemia and bleeding risks [16–19]. Therefore, the challenge of coordinating P2Y12 receptor inhibitor therapy has been intensively studied to prevent ischemic events with an acceptable bleeding risk in patients with ACS. Three different methods of de-escalation have recently been investigated: unguided de-escalation, platelet function testing-guided de-escalation, and genotype-guided de-escalation (Fig. 1).
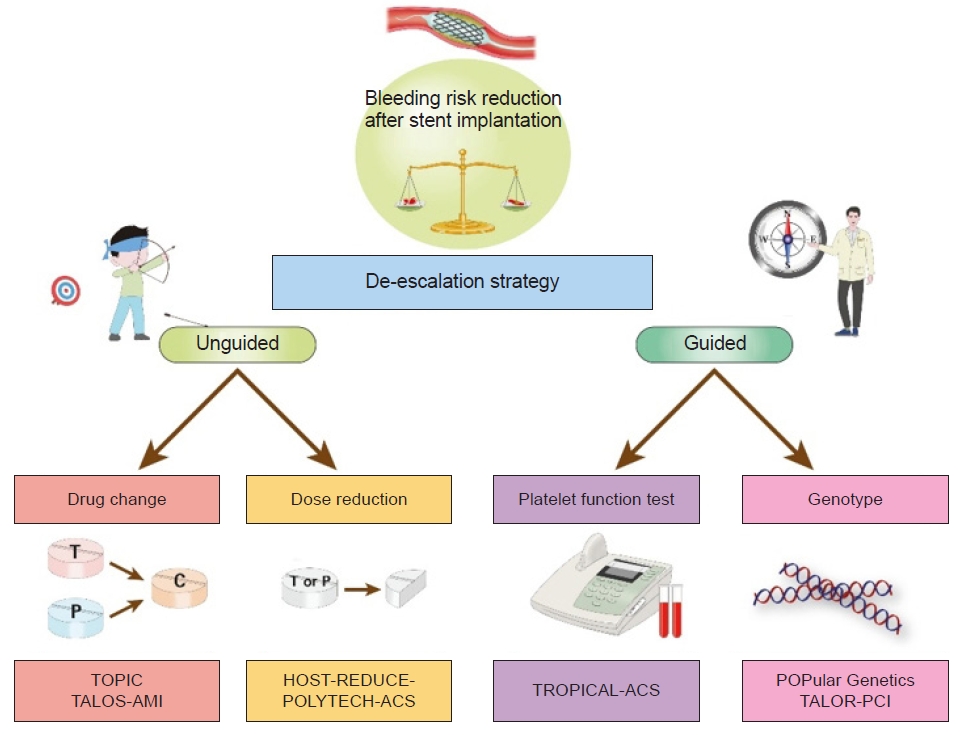
Different methods of de-escalation strategies of dual antiplatelet therapy in patients undergoing percutaneous coronary intervention for acute coronary syndrome. TOPIC, Timing of Platelet Inhibition after Acute Coronary Syndrome; TALOS-AMI, Ticagrelor versus Clopidogrel in Stabilized Patients with Acute Myocardial Infarction; HOST-REDUCE-POLYTECH-ACS, Harmonizing Optimal Strategy for Treatment of Coronary Artery Diseases-Comparison of Reduction of Prasugrel Dose or Polymer Technology in Acute Coronary Syndrome Patients; TROPICAL-ACS, Testing Responsiveness To Platelet Inhibition On Chronic Antiplatelet Treatment For Acute Coronary Syndromes; POPular Genetics, CYP2C19 Genotype-Guided Antiplatelet Therapy in ST-Segment Elevation Myocardial Infarction Patients — Patient Outcome after Primary Percutaneous Coronary Intervention; TAILOR-PCI, Tailored Antiplatelet Initiation to Lesson Outcomes Due to Decreased Clopidogrel Response After Percutaneous Coronary Intervention.
UNGUIDED P2Y12 INHIBITOR DE-ESCALATION
The Timing of Platelet Inhibition after Acute Coronary Syndrome (TOPIC) trial investigated unguided de-escalation from prasugrel or ticagrelor to clopidogrel and evaluated long-term treatment with aspirin and clopidogrel (de-escalation of DAPT) compared with aspirin and a potent P2Y12 inhibitor (unchanged DAPT) after 1 month of initial treatment with aspirin and a potent P2Y12 inhibitor for ACS [8]. Patients who were event-free for 1 month after ACS were randomized to de-escalation of DAPT (n=323) or unchanged DAPT (n=323). The risk of the combined primary endpoint, defined as a composite of death, urgent revascularization, stroke, and Bleeding Academic Research Consortium (BARC) ≥2 bleeding dropped by half in patients with de-escalation of DAPT therapy (26.3% vs. 13.4%; hazard ratio [HR], 0.48; 95% confidence interval [CI], 0.34–0.68). While there was no significant difference in ischemic endpoints between the groups (11.5% vs. 9.3%; HR, 0.80; 95% CI, 0.50–1.29), BARC ≥2 bleeding was significantly lower in the de-escalation group (14.9% vs. 4.0%; HR, 0.30; 95% CI, 0.18–0.50). All BARC and Thrombolysis in Myocardial Infarction (TIMI) minor bleeding also favored the de-escalation group, while TIMI major bleeding was not significantly different between the groups. The results of the TOPIC trial seem promising and pose a challenge to the current DAPT guidelines, but the single-center nature of the study and the small sample size warrant future larger trials in this field.
The Harmonizing Optimal Strategy for Treatment of Coronary Artery Diseases-Comparison of Reduction of Prasugrel Dose or Polymer Technology in Acute Coronary Syndrome Patients (HOST-REDUCE-POLYTECH-ACS) trial, which evaluated the prasugrel-based de-escalation of DAPT after PCI in patients with ACS, compared unguided prasugrel de-escalation (5 mg daily) versus standard-dose prasugrel (10 mg daily) 1 month after ACS patients underwent PCI [9]. Net adverse clinical events, defined as a composite of all-cause death, myocardial infarction (MI), stent thrombosis, repeat revascularization, stroke, and BARC ≥2 bleeding, occurred in 82 patients (7.2%) in the de-escalation group and 116 patients (10.1%) in the conventional group. Prasugrel dose de-escalation showed a significant risk reduction of net adverse clinical events compared with conventional-dose therapy (HR, 0.70; 95% CI, 0.52–0.92; P=0.012), which was mainly driven by a lower risk of BARC 2 bleeding, the only individual component of the primary endpoint that showed a significant difference. There was no significant difference in major bleeding (BARC ≥3) between the groups. The promising results of that trial should be weighed against several considerations. Although only patients with ACS were included, half of them had low-to-moderate risk profiles. Further analysis is required to determine whether prasugrel de-escalation is beneficial even in high-risk patients. In addition, the trial included only East Asian patients, who are known to be less prone to thrombosis and more prone to bleeding. Whether the findings are reproduceable in European and North American patients is uncertain.
The Ticagrelor versus Clopidogrel in Stabilized Patients with Acute Myocardial Infarction (TALOS-AMI) trial, which analyzed unguided de-escalation from ticagrelor to clopidogrel in stabilized patients with acute myocardial infarction undergoing PCI, randomized 2,697 Korean acute MI patients (STEMI, 54%) who had all received 1 month of ticagrelor and aspirin with no events to either de-escalation with aspirin and clopidogrel (n=1,349) or continued ticagrelor and aspirin (n=1,348) [10]. At 1 year, the risk of cardiovascular death, MI, stroke, or BARC ≥2 bleeding was almost halved in patients in the de-escalation group compared with the continued group (4.6% vs. 8.2%; HR, 0.55; 95% CI, 0.40–0.76), which was mainly driven by less bleeding (3.0% vs. 5.6%; HR, 0.52; 95% CI, 0.35–0.77). There was no significant difference in ischemic events between the groups (2.1% vs. 3.1%; HR, 0.69; 95% CI, 0.42–1.14). Per-protocol and subgroup analyses were consistent with the primary results. Like the HOST-REDUCE-POLYTECH-ACS trial, the TALOS-AMI trial only included East Asian patients, so its generalizability to Western populations is unclear.
PLATELET FUNCTION TESTING-GUIDED P2Y12 INHIBITOR DE-ESCALATION
The Testing Responsiveness To Platelet Inhibition On Chronic Antiplatelet Treatment For Acute Coronary Syndromes (TROPICAL-ACS) trial evaluated guided de-escalation of antiplatelet treatment in patients with acute coronary syndrome undergoing percutaneous coronary intervention, testing a guided de-escalation strategy based on platelet function tests in ACS patients undergoing PCI [11]. Patients were randomized to de-escalation of antiplatelet therapy (n=1,304) or continued prasugrel for 12 months (n=1,306). In the de-escalation group, patients received prasugrel for 1 week, then clopidogrel for 1 week at which time platelet function testing was performed. Patients without high platelet reactivity (HPR) continued clopidogrel, whereas those with HPR were switched to prasugrel. The rate of cardiovascular death, MI, stroke, or BARC grade ≥2 bleeding was 7.3% in the guided de-escalation group and 9.0% in the control group (P=0.0004 for noninferiority). The rates of BARC grade ≥2 bleeding and composite ischemic outcomes did not significantly differ between the groups. A prespecified analysis of the TROPICAL-ACS trial assessed the impact of age on clinical outcomes following guided de-escalation of antiplatelet treatment in ACS patients [12]. There was significant treatment interaction when age was analyzed as a continuous variable (P for interaction=0.02). The net clinical benefit was due to a reduction in major bleeding among younger patients receiving de-escalation therapy.
GENOTYPE-GUIDED DE-ESCALATION
Clopidogrel is a prodrug that is converted to its active metabolite in the liver via two oxidative steps that involve the cytochrome P450 superfamily enzyme system, of which the CYP2C19 enzyme is the most important component. Patients who have a loss of function (LOF) variant of the CYP2C19 gene are unable to fully metabolize the prodrug into the active metabolite, reducing its effectiveness and increasing the risk of ischemic events. The most common LOF alleles are CYP2C19*2 and CYP2C19*3. Previous studies have shown that patients who carry these LOF alleles have a higher incidence of ischemic events when they take clopidogrel than those without the alleles [20,21]. It is unknown whether routinely genotyping for CYP2C19 LOF alleles and prescribing antiplatelet therapy based on the results can reduce the incidence of ischemic events.
The CYP2C19 Genotype-Guided Antiplatelet Therapy in ST-Segment Elevation Myocardial Infarction Patients — Patient Outcome after Primary Percutaneous Coronary Intervention (POPular Genetics) trial evaluated a genotype-guided strategy for selection of a P2Y12 inhibitor compared to standard DAPT with ticagrelor or prasugrel [13]. Patients undergoing primary PCI for STEMI were randomized to a genotype-guided strategy for selection of an oral P2Y12 inhibitor (n=1,242) versus standard therapy with ticagrelor or prasugrel (n=1,246). In the genotype-guided group, patients with LOF received either ticagrelor (97%) or prasugrel (3%). Otherwise, patients received clopidogrel. The genotype-guided strategy was noninferior to standard care with respect to the primary composite endpoint of all-cause death, MI, definite stent thrombosis, stroke, and PLATelet inhibition and patient Outcomes (PLATO) major bleeding (5.1% vs. 5.9%; P for noninferiority <0.001). Additionally, genetic testing was associated with less major or minor bleeding (9.8% vs. 12.5%; HR, 0.78; 95% CI, 0.61–0.98; P=0.04). There were no significant differences between the cohorts regarding any other thrombotic secondary outcomes. The results were maintained in both per-protocol and sensitivity analyses. The use of a genotype-guided de-escalation strategy to select P2Y12 inhibitors has been proven to be beneficial by reducing bleeding risk without increasing ischemic events in STEMI patients undergoing primary PCI.
The Tailored Antiplatelet Initiation to Lesson Outcomes Due to Decreased Clopidogrel Response After Percutaneous Coronary Intervention (TAILOR-PCI) study tested the hypothesis that altering antiplatelet therapy based on CYP2C19 LOF status would lead to improved outcomes [22]. Patients undergoing PCI for stable or unstable angina were randomized to a genotype-guided strategy (n=2,652), in which patients without CYP2C19 LOF alleles received clopidogrel and patients with the LOF alleles received ticagrelor or prasugrel. The standard therapy (n=2,650) group received clopidogrel without prospective genotyping. The primary analysis cohort, based on a prespecified analysis plan, consisted of 946 patients in the conventional group and 903 patients in the genotype-guided group, who were identified as being carriers, at 12 months. Among patients who carried the genetic variant, the primary endpoint occurred in 4.0% of the genotype-guided group, compared with 5.9% in the conventional group (HR, 0.66; 95% CI, 0.43–1.02; P=0.056). No significant difference was seen in the safety endpoint of TIMI major bleeding or minor bleeding (1.9% vs. 1.6%, respectively). A prespecified sensitivity analysis for the primary endpoint found a 40% reduction for cumulative primary endpoint events that occurred during the study period (95% CI, 0.41–0.89; P=0.011). Therefore, when the time to multiple recurrent events was considered, there was a possible benefit to a genotype-guided strategy to identify patients with LOF.
CONCLUSIONS
With improvements in the understanding of the prognostic relevance of bleeding events in ACS patients with PCI, as well as the safety and efficacy of DESs, clinicians’ focus has shifted more towards preventing bleeding events. Although current guidelines do not recommend elective de-escalation of antiplatelet therapy, there is a growing body of evidence supporting a de-escalation strategy for antiplatelet therapy in ACS patients undergoing PCI. The most important consideration is to identify patient groups that might benefit more from de-escalation of potent antiplatelet therapy.
Notes
Ethical statements
Not applicable.
Conflicts of interest
The author has no conflicts of interest to declare.
Funding
None.